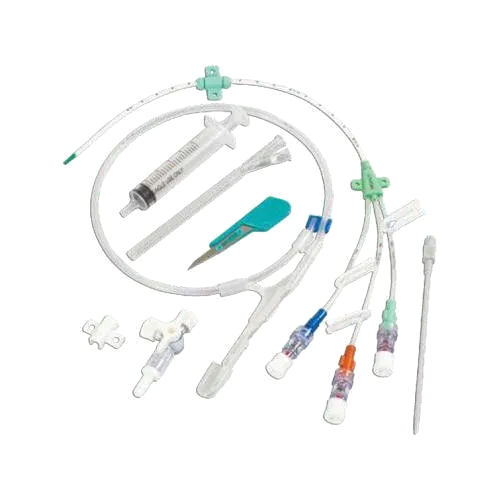
Epidural Catheter
Product Details:
- Equipment Type Epidural Catheter
- Material Plastic
- Condition New
- Use Hospital
- Color White
- Click to View more
Epidural Catheter Price And Quantity
- 50 Piece
- 500 INR/Piece
Epidural Catheter Product Specifications
- Hospital
- Plastic
- White
- New
- Epidural Catheter
Epidural Catheter Trade Information
- 10000 Piece Per Week
- 4 Days
Product Description
Product details
|
Available Length |
110 cm |
|
Size |
Medium |
|
Shape |
Straight-Single |
|
Brand |
Portex & Mercury |
|
Disposables |
Yes |
|
Material Type |
Plastic |
|
Type |
Central Venous |
Frequently Asked Questions
Q: An epidural catheter is what?
A: A tiny, thin tube called an epidural catheter is inserted into the epidural space, which is the region around the spinal cord and spinal fluid. It is used to deliver medications, such as local anaesthetics, to relieve pain in particular body parts.
Q: How is a catheter for an epidural inserted?
A: A medical expert must first implant a needle into the epidural area and then thread a tiny catheter through the needle to place an epidural catheter. The catheter is then left in place after the needle has been removed. In order to keep the catheter from moving, it is often fastened to the patient's skin.
Q: What function does an epidural catheter serve?
A: An epidural catheter is primarily used to deliver pain-relieving medicines. It is frequently used for pain management in people with chronic diseases, like cancer or back pain, as well as during delivery and surgery.
Q: How long can a catheter for an epidural stay in place?
A: Depending on the particular circumstances and the doctor's discretion, an epidural catheter may be left in place for a variety of reasons. After surgery, it might be left in for a few days, but in other situations, such as chronic pain treatment, it might be used for a longer period of time.
Q: What dangers and issues can epidural catheters bring about?
A: Although epidural catheters are generally safe, there may be dangers and issues. These could include an infection at the site of insertion, haemorrhage, nerve damage, headaches, breathing issues, or even a spinal cord injury in rare cases. Before the procedure, the risks and advantages should be reviewed with a medical expert.
Q: With an epidural catheter in place, am I able to move or walk?
A: An epidural catheter is typically attached to a pump or syringe that administers drugs constantly or sporadically. Mobility may be restricted or reduced depending on the circumstances and medications being taken. It's important to abide by the movement and activity limits prescribed by your doctor.
Q: The epidural catheter is withdrawn in what way?
A: Epidural catheter removal is a straightforward procedure that is usually carried out by a medical expert. The catheter is carefully removed once it has been confirmed that it is no longer required. After that, the insertion site is cleansed and bandaged.